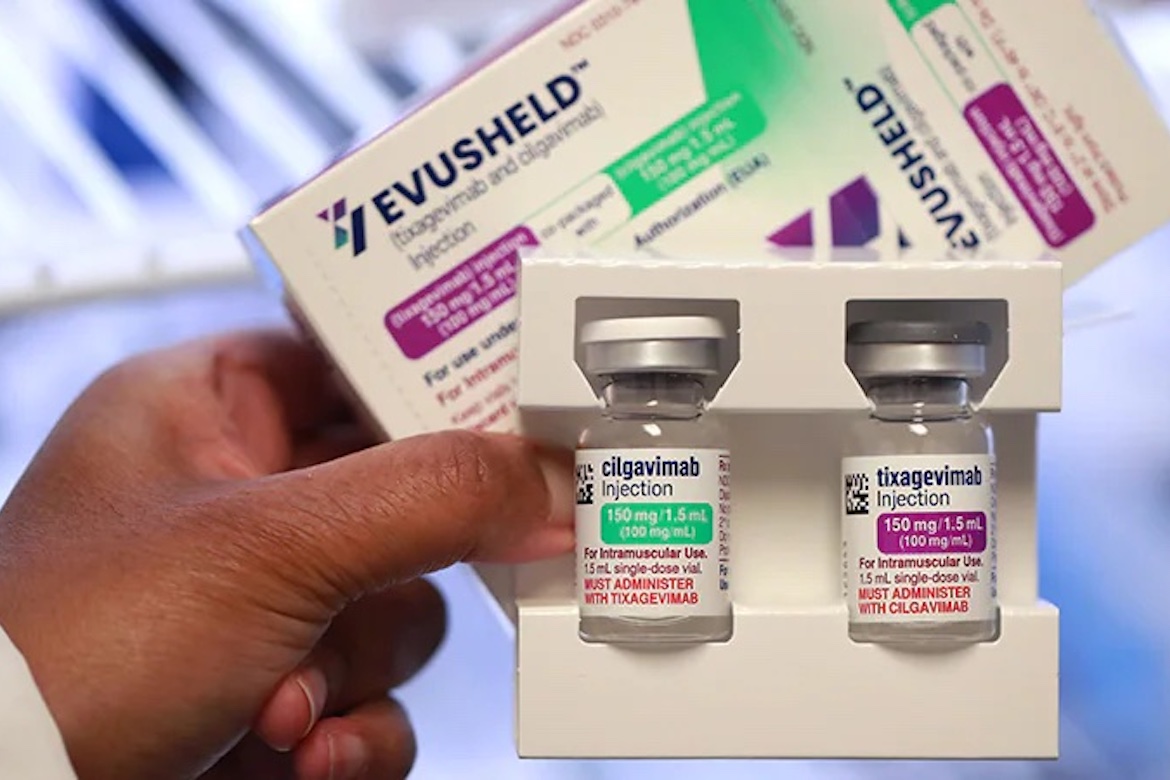
A new drug that helps prevent COVID-19 in people who are at a particular risk from the disease is now available in Greece.
On Thursday, the health ministry said Evusheld, a monoclonal antibody medicine developed by AstraZeneca, has been approved by the European Medicines Agency.
The medicine is approved for adults and children aged over 12, that weigh at least 90 pounds, who have compromised immune systems and are also at risk from the COVID vaccine.
Eligible recipients must also be completely clear of the coronavirus and have not been infected for at least three months.
The application for the two-injection dose of Evusheld needs to be submitted by the patient’s doctor, along with evidence supporting why he or she needs the medicine instead of a vaccine.
Meanwhile, COVID vaccines that have been updated to protect people against hospitalization and death from the Omicron variant are expected to receive approval by the end of the month.
Health Minister Thanos Plevris told Skai radio on Friday that the new vaccines will be available to the public by the fall, provided that they get approved.
Plevris said the vaccines will be further updated at a later date to include protection against the subvariants BA.4 and BA.5.
Is The Pappas Post worth $5 a month for all of the content you read? On any given month, we publish dozens of articles that educate, inform, entertain, inspire and enrich thousands who read The Pappas Post. I’m asking those who frequent the site to chip in and help keep the quality of our content high — and free. Click here and start your monthly or annual support today. If you choose to pay (a) $5/month or more or (b) $50/year or more then you will be able to browse our site completely ad-free!
Click here if you would like to subscribe to The Pappas Post Weekly News Update